Hello World 1
Welcome to Elementor


ReGEN Global Logistics delivers time-critical and regulated logistics solutions designed for environments where precision is non-negotiable. Our operating model is built around control, visibility, and accountability – ensuring critical cargo moves safely, compliantly, and without compromise.
We support organizations operating in regulated and mission-critical environments with a consistent, GDP-derived operating model that scales internationally while maintaining purpose-built oversight.
ReGEN Global Logistics provides specialized logistics solutions for pharmaceutical, biotech, clinical research and healthcare organizations operating in regulated and time‑sensitive environments. Our approach emphasizes structured execution, risk control and regulatory alignment—supporting supply chains where consistency and documentation are as critical as speed.

Time-critical logistics and emergency air freight across Canada and internationally

NFO logistics for urgent, life-science emergency transport

Pharmaceutical cold chain logistics and 24/7 cold chain intervention

Clinical trial supply chain management

Advanced therapy logistics for cell and gene therapies

Controlled substance and medical device logistics

Door-to-door white-glove delivery

First mile and last mile logistics with full chain-of-custody
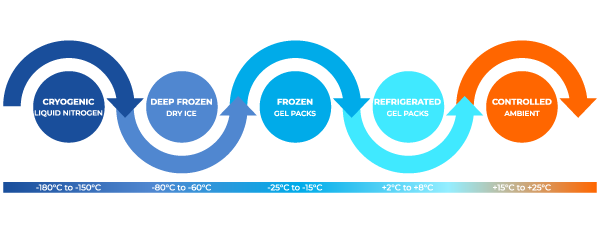
We offer a complete range of validated, temperature-controlled packaging solutions, precisely covering the critical range from -150°C (Cryogenic) to +25°C (Controlled Ambient).


Seamless logistics across all major global markets.

GDP-compliant, qualified handling facilities globally.

Constant oversight for global shipments and critical milestones.

Adherence to GDP and IATA standards for air and road transport.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.

We offer passive and active temperature‑controlled solutions designed to maintain common bands such as refrigerated 2°C–8°C, controlled ambient 15°C–25°C and ultra‑low or cryogenic ranges (e.g., –75°C or lower) when required. We can integrate real‑time monitoring and provide excursion reports in line with GDP guidelines.
Our quality system is aligned with GDP principles, focusing on qualified equipment, documented procedures, risk assessment and continuous monitoring. We use validated packaging, calibrate sensors, train staff and maintain audit‑ready documentation.
Our time‑critical logistics services include Next‑Flight‑Out and on‑board courier options with 24/7 operations and live monitoring. We follow predefined response protocols and immediate escalation to minimize delays.
We provide regulated logistics solutions for pharmaceuticals, biotechnology, clinical research, medical devices, diagnostic equipment, hospitals and labs, and direct‑to‑patient programs.
When deviations occur, we document each event and calculate Mean Kinetic Temperature in accordance with USP <1079.2>. We treat excursions as nonconformities, investigate root causes and implement corrective actions.